Search from website
Search from website
Home CRDMO Services Protein production
From simplest of human IgG’s to afucosylated bispecific antibodies up to recombinant growth factors — we have expressed and purified them all!
Icosagen provides custom recombinant protein production services, using proprietary mammalian cell-based QMCF Technology with animal-free components.
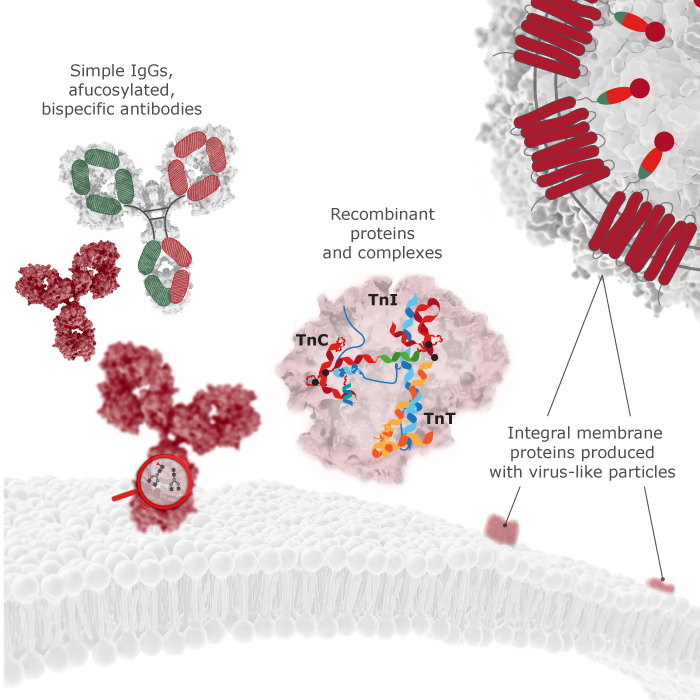
Our QMCF Technology is a fast and reliable transient mammalian expression system for recombinant protein production with high yields up to 1g/L.
QMCF utilizes genetic modifications that increase the expression vector copy-number while ensuring their even distribution during cell division, guaranteeing stable expression levels.
Produced recombinant proteins are suitable for various types of pre-clinical research from structural biology to drug discovery as well as for commercial diagnostic assays.
Production volumes range from 96-well plate to 50-litre scale. A wide array of current analytical methods and equipment used throughout the production process ensures consistent highest quality.
A variety of protein purification capabilities are established, ranging form affinity-based purifications to tagless proteins.

We produce and purify bispecific and multispecific antibodies, and similar antibody-like molecules for therapeutic and diagnostic purposes. We can start from in-house discovery project to identify target candidates, by employing in vivo and in vitro antibody discovery platforms. As a more straightforward approach, we can produce and purify antibodies based on electronic sequences provided by our clients.
Utilizing different custom-tailored protein purification methods, we adapt purification schemes for each molecule. Coupled with thorough panel of analytics, detailed insights into each molecule will be provided, supporting informed decision-making during every stage of the project.


With our technology, VLPs can be produced in HEK293-based QMCF
cell lines and purified by precipitation and/or density gradient fractionation.
Transmembrane proteins, such as GPCRs, ion channels, and transporters are notoriously difficult to purify since they require the cell membrane lipid bilayer to maintain their native, active 3D-fold. The discovery of antibodies against such transmembrane proteins proved therefore very difficult in the past. GPCRs for example, are the largest class of cell-surface proteins that are targeted by almost 40% of all marketed drugs, but these are all small molecule drugs.
Icosagen has overcome this challenge by developing a proprietary pseudotyped VLP technology platform to conduct antibody discovery projects in-house on transmembrane proteins. We can produce transmembrane proteins in the context of virus-like particles (VLPs), using our QMCF mammalian expression platform. The co-expression of a viral envelope or capsid protein together with the transmembrane protein results in the self-assembly of pseudotyped VLPs, which are enriched with the membrane protein of interest. These pseudotyped VLPs serve for immunization and are frequently used in antibody screening, vaccine development, and receptor function studies.
Icosagen has a long-standing partnership with ProBioGen to use its GlymaxX® technology for generating fully afucosylated and therefore ADCC-enhanced antibodies. We offer rapid, multi-parallel, transiently produced ADCC-enhanced mAbs to assess cell-killing potency enhancement effects on any antibody.
GlymaxX® is universally applicable, guarantees over 95% afucosylation and can be rapidly applied into existing production cell lines or to new antibody development projects. Production timelines and final yields are identical for both regular and afucosylated antibodies.
Therapeutic antibodies with low fucose content have shown 10-100 fold higher antibody-dependent cellular cytotoxicity (ADCC) and thus drastically higher target cell killing potency. The degree of afucosylation levels can even be adjusted, hence the cell killing potency as well.



